> HAA := R -> > e^2/(4*Pi*epsilon0*a0)*(exp(-2*R/a0)*(1+a0/R)-a0/R-1/2);
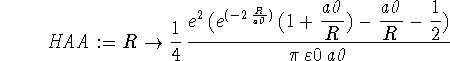
> HAB := R -> > -e^2/(4*Pi*epsilon0*a0)*(SAB(R)/2+exp(-R/a0)*(1+R/a0));
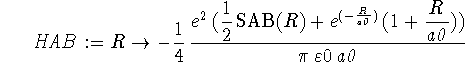
> SAB := R -> exp(-R/a0)*(1+R/a0+(R/a0)^2/3);
![]()
> Ep := R -> (HAA(R)+HAB(R))/(1+SAB(R));
![]()
> Em := R -> (HAA(R)-HAB(R))/(1-SAB(R));
![]()
> assume(a0>0); > limit(Ep(R),R=infinity);
![]()
> limit(Em(R),R=infinity);
![]()
This is exactly the ground-state energy of a hydrogen atom. This
makes sense because as ![]() , a hydrogen molecular ion
should separate into a hydrogen atom and a proton.
, a hydrogen molecular ion
should separate into a hydrogen atom and a proton.
> Veffp := R -> Ep(R) + e^2/(4*Pi*epsilon0*R);
![]()
> Veffm := R -> Em(R) + e^2/(4*Pi*epsilon0*R);
![]()
> e := 1.6022e-19;
![]()
> epsilon0 := 8.8542e-12;
![]()
> a0 := 5.292e-11;
![]()
> plot({Veffp,Veffm},0..7*a0,-3e-18..1e-18);
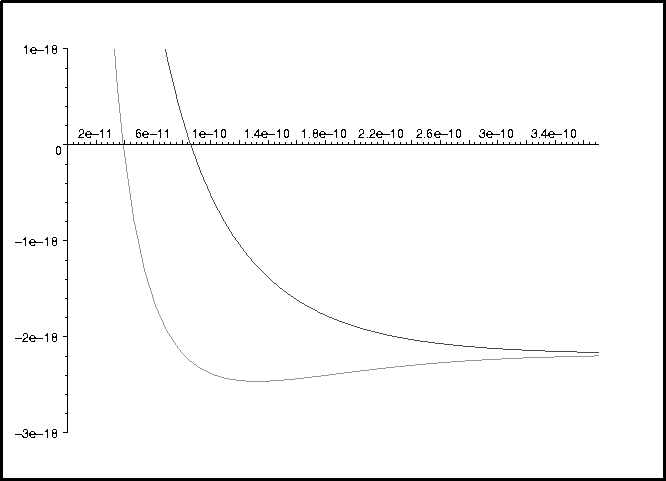
To figure out which curve is which:
> evalf(Veffp(1e-10));
![]()
> evalf(Veffm(1e-10));
![]()
The bonding state is represented by the + effective potential energy curve (Veffp).
> Req:=fsolve(diff(Veffp(R),R)=0,R,5e-11..1.5e-10);
![]()
> k := evalf(subs(R=Req,1/2*diff(Veffp(R),R$2)));
![]()
> mH := 1.0078e-3/6.0221e23;
![]()
> mu := mH/2;
![]()
> omega0 := sqrt(k/mu);
![]()
> hbar := 6.6261e-34/(2*Pi);
![]()
> E0 := evalf(1/2*hbar*omega0);
![]()
> evalf(limit(Veffp(R),R=infinity)-Veffp(Req)+E0);
![]()