> psi2s := (r,theta,phi) -> 1/8*sqrt(2/(Pi*a0^3))*(2-r/a0)*exp(-r/(2*a0));
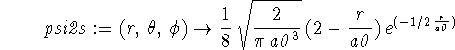
> psi3s := (r,theta,phi) -> 1/243*sqrt(3/(Pi*a0^3))*(27-18*r/a0+2*(r/a0)^2) *exp(-r/(3*a0));
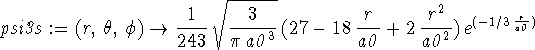
Maple will need to know the sign of a0 to solve the problems in
this assignment.
> assume(a0>0);
In the 2s state,
> int(int(int( evalc(conjugate(psi2s(r,theta,phi))*r*psi2s(r,theta,phi)) *r^2*sin(theta),theta=0..Pi),phi=0..2*Pi),r=0..infinity);
![]()
Note that the use of
evalc() and conjugate() are not
strictly required in this and the following
case since the wavefunctions are real.
In the 3s state,
> int(int(int( evalc(conjugate(psi3s(r,theta,phi))*r*psi3s(r,theta,phi)) *r^2*sin(theta),theta=0..Pi),phi=0..2*Pi),r=0..infinity);
![]()
As discussed in introductory chemistry courses, the shells get bigger
(the electrons spend more time away from the nucleus) as the principal
quantum number increases. The increase in average distance from the
nucleus ( ![]() ) reflects this.
) reflects this.
-
Let's start with the 3s radial probability density (r.p.d.):
> rpd3s := r -> N3s*r^2*(27-18*r/a0+2*(r/a0)^2)^2 *exp(-2*r/(3*a0));
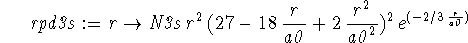
Here,N3sis a normalization factor which we must determine:
> solve(int(rpd3s(r),r=0..infinity)=1,N3s);

> N3s:=";
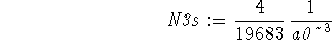
Now, for the
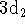 state (
state ( 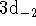 has an identical r.p.d.):
has an identical r.p.d.):
> rpd3d2 := r -> N3d2*r^2*(r/a0)^4*exp(-2*r/(3*a0));
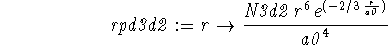
> solve(int(rpd3d2(r),r=0..infinity)=1,N3d2);

> N3d2:=";
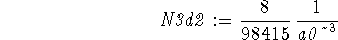
-
For the 3s state:
> plot(subs(a0=1,rpd3s(r)),r=0..40);
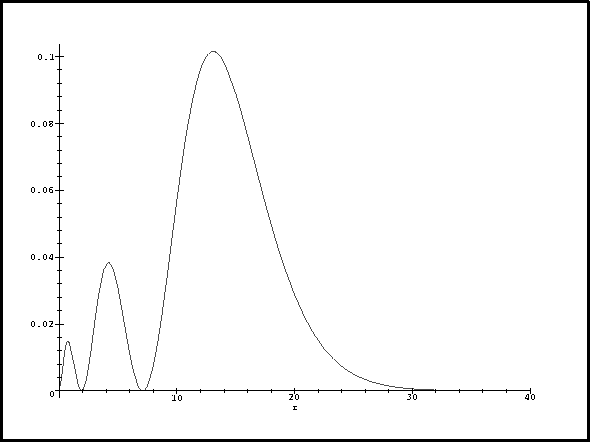
Note that I chose the upper limit of the plot command large enough that I could see the whole function.For the
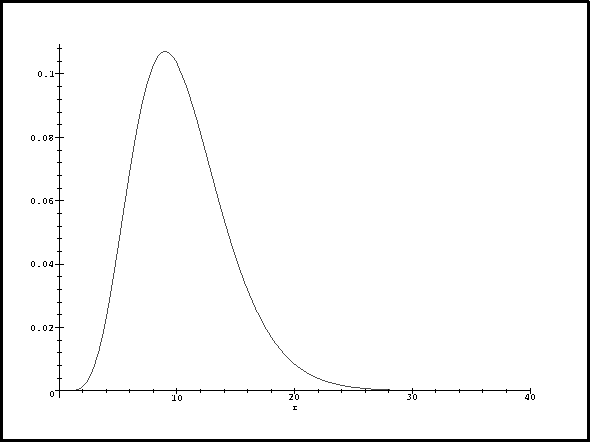
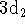 state:
state:
> plot(subs(a0=1,rpd3d2(r)),r=0..40);
- We want to find the places where the 3s r.p.d. is zero:
> solve(rpd3s(r)=0,r);
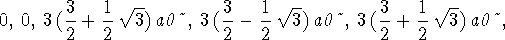
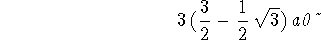
The radial nodes are
> rn1:="[3];
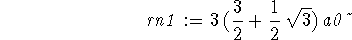
> rn2 := ""[4];
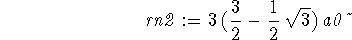
> rn0:=0;

-
rn1 is the last node.
> int(rpd3s(r),r=rn1..infinity);
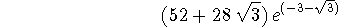
> evalf(");