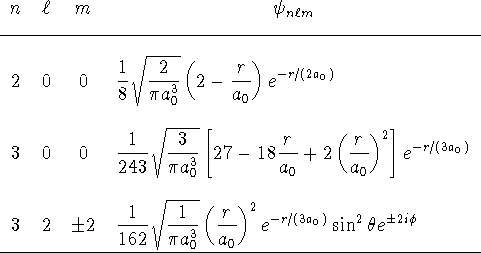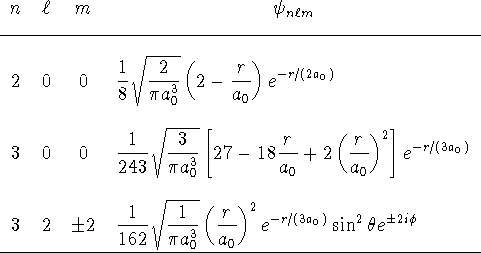Up: Back to the Chemistry 3730 assignment
index
Chemistry 3730 assignment 8
Due: Friday, Oct. 31, 10:00 a.m.
Use the following table of hydrogenic wavefunctions to answer the
questions:
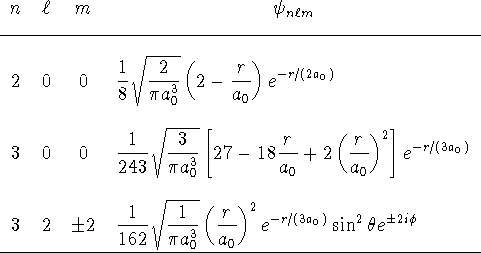
- Calculate
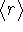 in the 2s and 3s states.
Combining these results with the 1s result computed in class
(
in the 2s and 3s states.
Combining these results with the 1s result computed in class
( 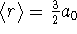 ),
comment on the results with respect to a simple shell model of
the atom.
),
comment on the results with respect to a simple shell model of
the atom.
Maple hint: You will have to use assume(a0>0);
- Work out the normalized
3s and
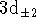 radial
probability distributions.
radial
probability distributions. - Sketch these distributions.
- Radial nodes are points where the radial probability
distribution is zero. Locate the radial nodes of the 3s
state.
Maple hint: Use solve() to find the nodes. Maple
may report redundant solutions (i.e. it may print out
the same root more than once).
- Compute the probability that the electron will be found
between the last node and infinity in the 3s state.
Report your answer to four significant figures.
Hint: If you have a normalized r.d.f., you do not need
to use the full wavefunction.
Marc Roussel
Wed Oct 22 12:57:57 MDT 1997