


Up: Back to the Chemistry 3730 assignment
index
Chemistry 3730 assignment 4
Due: Wednesday, Oct. 1, 10:00 a.m.
Suppose that a particle in a box is subjected to a perturbing potential
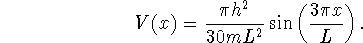
- Compute the energy shift due to the perturbation for n = 1, 2 and 3.
- Which of the shifts is greatest? Give a qualitative explanation
of why this should be so. It may be helpful to plot or sketch
the probability density and perturbation.
- Draw a diagram showing the original (unperturbed) energies and
the perturbed energies on the same scale. (I suggest you do
this by hand.) Your energy axis is probably most conveniently
labeled in units of
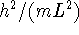 .
. - Would the wavelength of the photon required to cause a transition
from the ground state to the first excited state increase or
decrease after adding the perturbation?
Marc Roussel
Fri Sep 26 11:38:39 MDT 1997
![]()